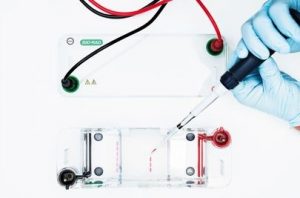
This firm is trying to grow red blood cells in the lab
This firm is trying to grow red blood cells in the lab
The NHS Blood and Transplant facility in Bristol, where donated blood is screened. A British team called Novosang wants to render this process obsolete
Credit Greg White
Monday 28 December 2015
Back In 2007, the US Defense Advanced Research Projects Agency (Darpa) issued an open call for a project. It required applicants to make a bioreactor that could produce an unlimited supply of blood from stem cells. The machine would be 1.3m3, roughly the size of 2.5 domestic washing machines, and hardy enough for it to withstand being parachuted into a battlefield and still function. The output was to be automatically packaged in standard blood bags. Darpa was offering a grant of $49 million (£32m). And the machine had to be completed within 27 months.
One of the teams to apply was a consortium of British academic groups and blood services. The senior figures in the group included Willy Murphy, medical and scientific director of the Irish Blood Transfusion Service; Dave Anstey, a researcher at NHS Blood and Transplant; Marc Turner, medical director of the Scottish National Blood Transfusion Service; and Joanne Mountford, a biomedical stem-cell researcher at the University of Glasgow. “The majority of us in the consortium were already working on adult stem cells but we hadn’t put the idea together of using our technology to make red blood cells,”
Mountford says.
“Of all the tissues that you might have wanted to make from stem cells, you would have argued that red blood cells were not one at all, because we have a perfectly good supply of them already. Why reinvent that wheel? What we really need is neurons, what we really need is liver cells, because we don’t have sources of those. But we started to realise that the idea of making blood in a machine – essentially a slot machine for blood – actually made sense. It was always going to be a massive challenge biologically and from an engineering point of view, but we could see that it might be doable, at a cost.”
The British consortium, Novosang, wrote a 48-page report about how it would build such a machine, which it submitted to Darpa on February 14, 2008. As with all the other teams that applied, its application was rejected.
Joanne Mountford prepares a sample for flow cytometry in her lab at the University of Glasgow
Credit Greg White
“They had some very specific requirements which were, quite frankly, quite impossible and bizarre,” Mountford tells WIRED at the University of Glasgow. She’s in her forties, has thick, dark hair and smiles after her lengthy scientific explanations. Novosang always felt that it was never going to get Darpa’s grant – the project was simply too pie in the sky. But what Mountford and the other British researchers came to realise was that manufacturing high quantities of blood from stem cells could actually be a solution to a very complex problem. Every year around the world, there are 100 million blood transfusions (around 2.2 million in the UK) – taking blood from a donor and giving it to a patient – saving millions of lives. But this simple therapy comes with significant potential complications. Although most blood services can filter donated blood against infectious diseases, there remains a risk of undetected pathogens slipping through. In the 90s, concerns were raised about undetected HIV. In 2013, British researchers estimated that one in 2,000 people in the UK carried the variant Creutzfeldt-Jakob disease. At the time there were three cases connected to transfusions – and there was no test for it. “We don’t know what that disease will be that we can’t screen against, so it will always be a retrospective action to put it right,” Mountford says. Furthermore, although national blood services in developed countries have sufficient reserves, in developing nations it’s precarious. In the developing world, for instance, more than 150,000 women die every year from post-partum haemorrhage because there is no blood available for transfusion.
As a result, there is still a global need for clean blood. Mountford and her fellow Novosang scientists understood that an unlimited supplied of lab-grown, infection-free human blood could potentially save millions of lives. Soon after being rejected for the Darpa grant, they stripped out the most outlandish and unsubstantiated aspects of their report and submitted it elsewhere. Later that year, they received a grant from the Wellcome Trust and the Scottish Funding Council. To date, they have raised more than £12 million. The plan: to make a sustainable source of lab-grown blood.
Donated blood needs to be prepared before being sent to hospitals and clinics. A unit bag of blood, usually 430ml, is hung on a drip, where a filter removes all the white cells and clot-forming platelets. What remains is then centrifuged and the two remaining components – red blood cells and plasma – are separated. After the plasma is removed, the red blood cells are then labelled according to blood type and tested for infection. These cells are then stored in blood banks, ready for transfusion. The red blood cells are the only component of the blood used in transfusions.
Red blood cells are about eight micrometres wide and rich in haemoglobin, a protein that binds to oxygen and gives them their colour. We have approximately 25 trillion red cells in our bodies at any given moment. They have an oval bi-concave shape and lack a nucleus, which makes them highly flexible. This is a useful feature as red cells need to squeeze through blood capillaries as narrow as two micrometres wide to reach other tissues, where they deliver oxygen and remove CO2 from our bodies.
Red cells circulate in the bloodstream for a period of around 120 days before they are digested by macrophages, a type of white blood cell, and cleared away by the liver. To replace them, new red blood cells are produced at a rate of two million every second from inside our bone marrow.
In the lab, Mountford wants to reproduce exactly what happens in the bone marrow when red blood cells are made. “We call them cultured red blood cells,” Mountford says. “The key is that they are not synthetic and this is not an artificial process – they are the exact same red blood cells we produce in the body. It’s just that they are grown in the laboratory. It’s a product as nature intended.”
Every cell in the body derives from a type of stem cell called pluripotent stem cell. Stem cells are, in a sense, the primordial cell from which every other cell is made as it matures through cellular differentiation. The maturation depends on the type of chemical and physical inputs in the surrounding environment. For instance, inside the bone marrow, where red blood cells are made, pluripotent stem cells first develop into a slightly more mature type of stem cell called a haematopoietic stem cell. This can give rise not only to red cells but to myriad other blood cells, such as T-lymphocytes and plasma cells. “We can isolate these haematopoietic cells and grow them into red blood cells in the lab,” Mountford says.
“The difficulty is they’re limited in their capacity to proliferate so we can only get a certain amount of red cells from any individual donation of bone marrow, so we would need to keep going back to donors. That’s not a practical solution for the future.”
In contrast, pluripotent stem cells can self-renew and proliferate indefinitely when grown in the lab. These cells are usually extracted from unused embryos from IVF treatments, with consent from the donors, and have the potential to differentiate indefinitely into any cells of the body. Of course, stem-cell research using embryonic cells has always been restricted due to ethical considerations regarding the use of embryos. However, those ethical issues were effectively circumvented in 2007 by Shinya Yamanaka, a Japanese biologist from the University of Kyoto, who was able to take mature skin cells – at a stage where they could no longer differentiate or divide – and, by adding four genes, turn them back into pluripotent stem cells.
In essence, Yamanaka rejuvenated the adult skin cells back to their primordial form so pluripotent cells could be turned into anything. “It seemed like alchemy at the time,” Montford says.
“It turned out to be the most incredible science I’ve seen in my career. He took cells which were terminally mature, fully differentiated, couldn’t divide, couldn’t turn into anything else, and he was able to revert them into a pluripotent nature so that they look and act identically to the human pluripotent stem cells from embryos.”
One year after Yamanaka’s discovery, Robert Lanza, an American stem-cell researcher and one of the first scientists to clone an early-stage human embryo, made red blood cells from human embryonic stem cells. One Sunday afternoon in the summer of 2008, Mountford made a similar breakthrough. She received an image sent by one of her research students. The picture showed a 2.5cm Eppendorf tube, a plastic lab tube used in centrifuges. The picture was fuzzy, but clear enough to show red fluid on the bottom of the tube. “It was the first picture we have of red cells made from pluripotent cells,” Mountford says. “It was nothing really, just 100,000 cells, around 8ml. And to get these 100,000 we had to put in about half a million cells, so from day zero to the end we were losing cells.”
Today, Mountford and her team can produce 2 x 1012 red blood cells, the number of red blood cells in a unit of blood used in transfusions, out of nine million stem cells. “It’s now sustainable and becoming a practical proposition, rather than having to have gazillions of cells at the beginning,” she says.
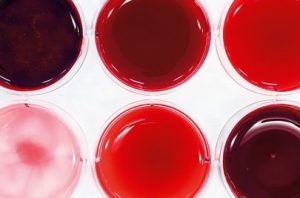
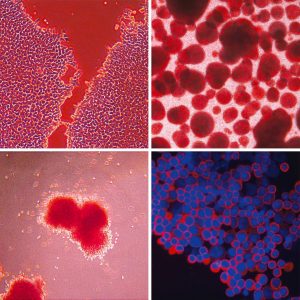
YOUR BLOOD UNDER THE MICROSCOPE
The four key stages in blood manufacture, which Novosang is emulating in the lab
When WIRED visits Joanne Mountford’s lab at the University of Glasgow, she guides us through labyrinthine corridors until we arrive at the cell-culture lab. Inside a small room, there’s a tissue-culture hood – an enclosed space designed to prevent contamination when handling cells. “Cells have to be handled in a sterile way,” Mountford says. “Also, if they were hazardous in any way, the user would be protected as well.” Next to the hood is an incubator, a humidified cabinet where stem cells grow inside tiny Petri dishes filled with a yellow liquid, a mixture of salts, sugars, amino acids and proteins. “These need feeding,” Mountford notes. “We feed them every day. We give them all the good stuff that they can eat and specific growth factors to stop the cells differentiating randomly. Cells will differentiate on their own, given half a chance.”
Mountford puts one of the dishes under the microscope, revealing a peninsular shape formed by thousands of cells on the plastic of the dish, separated by an empty area. Those, Mountford tells WIRED, are human embryonic stem cells in their early days. In a single dish there are about two million and, in theory, they can be turned into heart cells, liver cells, brain cells and, of course, blood cells. On top of this layer are bigger cells that seem to float. “Those cells are starting to differentiate spontaneously,” Mountford says. “That’s what we have to fight against. We have to stop them doing their own thing and get them to do what we want, which is [become blood] cells.”
Mountford produces another dish. “This is about day ten of the differentiation process,” she says. “They’re deciding if they want to be haematopoietic or not.” There are star-shaped cells stuck to the bottom of the dish and little balls of cells coming from them. “The stellate ones are vein cells, and haematopoietic cells are generated from them. They can turn into any kind of blood cell. We haven’t yet nudged them into being a red cell.”
Obtaining red blood cells from pluripotent induced stem cells remains a very complex problem. The pluripotent stem cells have a genetic programme that’s still very open and permissive, whereas a mature differentiated cell, such as a skin cell or a hair cell, has most of its DNA closed down. “The sequence has not changed, but the way it’s packaged is altered,” Mountford explains.
“In a mature cell, all of the options the cell doesn’t need are switched off. This expression was driven by the proteins and growth factors that were added in. A pluripotent stem cell, on the other hand, doesn’t have anything switched off yet. All the options are still open.”
Turning a pluripotent stem cell into a red blood cell takes about 31 days. Throughout that period the researchers need to follow a “recipe” with the right ingredients given to the stem cells at the right time and in the right doses. Finding the recipe is a haphazard task. “We just put things in, change the timings and see what happens,” Mountford says. “What we would ideally like to do is to be able to put them in a dish and then systematically work out what’s needed in what dose at what time. That’s the way things are starting to move. It’s basically suck-it-and-see for a lot of it.”
Luc Douay, a researcher at the Pierre and Marie Curie University in Paris, became in 2012 the first – and so far only – researcher to conduct a human clinical trial with red blood cells derived from stem cells. “He took adult stem cells, turned them into red cells and put them back into the same individual as proof of principle that this can work,” Mountford says.
“He found that red blood cells made from stem cells lasted longer in circulation than donated red cells.”
Today, Novosang includes researchers from the universities of Cambridge, Glasgow, Edinburgh, Bristol and Loughborough. It’s planning to run the first in-human trial in 2017, using red blood cells produced from adult stem cells. The objective will be to assess how manufactured red cells survive in comparison to donated cells. Ten volunteers will receive a transfusion of donated red cells and, six months later, a transfusion of manufactured red cells. “We’ve got all the expertise that’s required across different blood services to lead in this field,” says Nick Watkins, assistant director of research and development at NHS Blood and Transplant. The study will take about 18 months to run.
One of the side effects of regular transfusions is accumulation of iron in the liver and the heart. This is a problem that can be mitigated by lab-grown red blood cells. “The blood cells derived from stem cells survive longer than donated cells,” Watkins says. “If you’re a patient who requires regular transfusions, one of the biggest complications is iron overload. But if you increase the time between transfusions you can maybe reduce complications.”
Mountford agrees:
“In a normal bag of blood, every single red blood cell is a different age. One might be a day old, another 120 days old – the average lifespan of a red cell. The distribution of ages in a bag is random. But red cells grown in a lab will all be young and freshly produced, so the transfused blood will last longer in a patient.”
The challenge for Novosang is to manufacture red blood cells in sufficiently high quantities and in an economically sustainable way. A bag of blood produced using stem cells currently costs around £30,000 to make. In comparison, an average unit of blood at the NHS costs around £140 for about 450ml. “That’s without the infrastructure costs,” Mountford says. “When you cost in the blood services it’s significantly higher; I’m not sure that we have an absolute number.”
The other problem is to make enough. “When we get to clinical trials, we’re going to have to go up to 1,000-litre bioreactors,” Mountford says.
“If we’re ever going to produce the full UK annual requirement – 1018 cells – we’re looking at very large plants. We always hope that Grangemouth [Scotland] will stop petrol production. That would be good for us. It’s close.”
João Medeiros is WIRED’s science editor. He curates the R&D section.